| Summary: Nutrition and supplement brands face strict FDA, GMP, and EU rules, covering everything from labeling and batch traceability to multi-stage quality control, so manual paperwork quickly turns into errors, audit pain, and real business risk (fines, bans, recalls, and lost trust). This article explains how an ERP system centralizes data, automates documentation, and enables end-to-end batch tracking, quality checks, expiration control, and audit-ready records. It also shows how Odoo (especially its Quality and Document Management modules) can standardize processes, trigger quality alerts, and integrate production, warehousing, and delivery in one platform. |
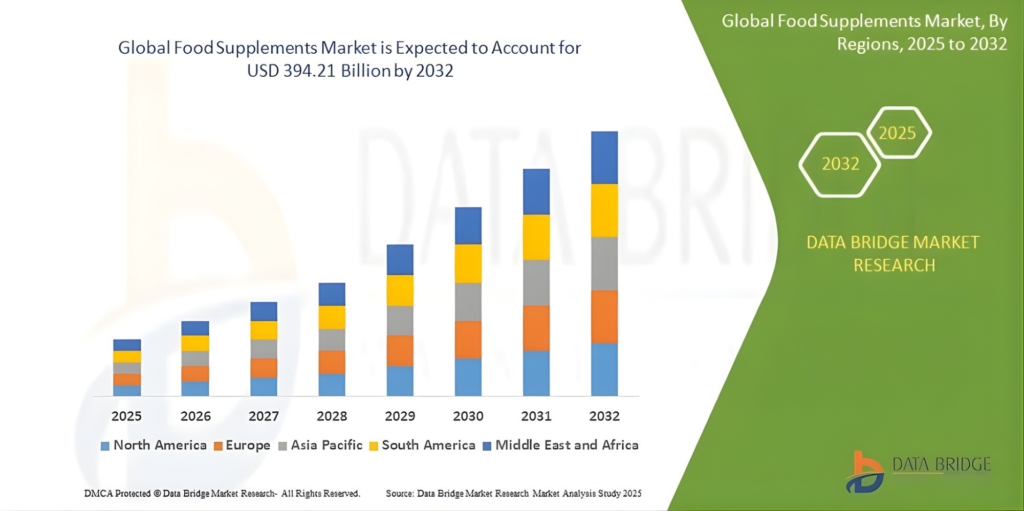
The nutrition and supplements industry is one of the most heavily regulated today. Regulatory compliance for nutrition brands can be a significant challenge, one that must be prepared for. The nutrition and supplements market is growing rapidly, and, according to Data Bridge Market Research, is expected to reach $394.21 billion by 2032. Market growth is driven by increased interest in natural medicine, preventive care, and greater consumer awareness of health issues.
Brands that fail to comply with regulatory standards suffer many negative consequences and have no chance to be competitive. Sales bans, fines, lawsuits and inspections, and, of course, loss of trust from partners and consumers are just some of the larger problems awaiting companies that fail to comply with regulations.
In this article, we will discuss regulatory requirements for nutrition companies and the main problems such brands face in today’s environment. We will also detail the importance of ERP systems and highlight the advantages of Odoo for regulatory compliance.
Key Regulatory Requirements in the Nutrition Sector
Ensuring nutrition products safety is a fundamental requirement of modern society and a key aspect of protecting public health. Different countries have their own regulations, and in some respects, they significantly differ from each other. For example, depending on the jurisdiction, the requirements for manufacturers and the level of control before a product enters the market vary. It is important to note that product labeling and product safety regulations for the nutrition industry are relevant regardless of the country, while implementation methods differ.

FDA
The FDA (Food and Drug Administration) of the US Department of Health and Human Services is responsible for regulating the quality of food, drugs, tobacco products, and cosmetics, as well as enforcing compliance with laws and standards in this area. The FDA regulates about 78% of food supplies in the US, which includes everything people eat, except for meat, poultry, and some egg products.
The main regulatory requirements of the FDA include:
- Proof of safety and efficiency. This includes clinical trials of the product lasting at least one year and involving at least 3,000 patients.
- Product labeling and packaging. Product labeling must include the name, manufacturer information, ingredients, and net weight. In addition, food products must include a nutrition facts table, and dietary supplement packaging must contain contact information for reporting side effects.
- Production standards. The FDA sets requirements for production facilities that call for the integrity and accuracy of information. Therefore, nutrition manufacturing software is extremely important.
GMP
GMP compliance (Good Manufacturing Practice) ensures that products are consistently manufactured in accordance with specifications and are safe and effective. GMP is a set of rules for the manufacture of high-quality medical products, namely medicines, dietary supplements, nutrition products, active ingredients, therapeutic and prophylactic agents, medical devices, and cosmetics. GMP certification guarantees high-quality standards at every production stage.
The main regulatory requirements of GMP include:
- Documentation and traceability. Each batch of products must be accompanied by a complete set of documents reflecting the production conditions and quality control results.
- Clearly defined processes. All stages of production must be documented in detail; this includes control and storage.
- Multi-stage quality control. This must be carried out at all stages of production, from the acceptance of raw materials to packaging and labeling.
- Staff training. All employees must undergo regular training on hygiene standards, working with equipment, and quality control methods.
- Premises and technical equipment. Production facilities must have equipment that is easy to clean, comply with sanitary and hygiene requirements, and maintain the necessary parameters.
- Validation and qualification. Critical equipment and processes are subject to validation and qualification procedures that confirm their compliance with standards.
- Change management. All changes to equipment or technological processes must be recorded, analyzed, and approved by authorized specialists.
EU Compliance
The requirements for nutrition production in the EU are extensive and include Regulation EC 1333/2008, Regulation (EC) No 178/2002, Regulation (EC) No 1331/2008, Regulation (EU) No 234/2011, Regulation (EU) No 562/2012, and Regulation (EU) 2019/1381. All nutritional supplements must be authorized and approved for use in the EU, and they should be based on technological necessity, safety assessment, and the absence of the possibility of misleading consumers.
The main regulatory requirements in the EU include:
- Food safety and nutrition supplements. All supplements must be on the list of supplements permitted in the EU.
- Labelling. Packaging must indicate the nutritional value of the product and the amounts of fat, sugar, and salt. The ingredients must be listed, allergens must be clearly identified, and the product name, expiration date, date of manufacture, and information about its origin must be indicated.
Laboratory Testing and Certification
Lab testing and certification in the nutrition industry are essential, as they confirm not only the quality but also the safety of the products. This is verified by many different procedures, and documentation confirms the product’s compliance with standards.
Laboratory tests are aimed at confirming product safety, determining its nutritional value, and ensuring compliance with standards. Such laboratory tests help identify the content of harmful substances, the product’s shelf life, and the compliance of the nutritional value labeling with the product’s actual composition.
Certification is usually carried out by accredited bodies and is based on the results of laboratory tests. This certification officially confirms that a product meets quality and safety requirements.
Typical Problems Faced by Nutrition Brands in the Current Environment
In the era of digital business transformation, companies that do not implement modern technologies face many challenges such as, regularly receiving fines, losing customer trust, and being unable to scale up to new markets. Read on to learn more about the most common problems faced by such nutrition brands.
Errors in Manual Documentation Flow
Document control is one of the most important components of regulatory compliance in the nutrition and supplements industry, and it encompasses various aspects. This includes the creation, approval, distribution, retrieval, and archiving of documents required by international quality standards. Despite the importance of this aspect, document control processes often prove ineffective during inspections. The most common reasons for this are the use of outdated versions and incorrect documentation.
When expanding a product line, for example, the number of documents that need to be controlled increases significantly. With such a huge volume of documents, manual documentation flow leads to duplication, errors, and contradictions between documents. In addition, a complex documentation hierarchy means that users are highly likely to refer to outdated instructions. And if the company also has a mixture of paper, electronic, and PDF files, the risk of using uncontrolled documents is extremely high.
Insufficient Transparency in Supply Chain
The supply chain is an important element of a nutrition business, and its lack of transparency leads to many problems and jeopardizes the company’s reputation. Lack of transparency increases the risk of disruptions, makes it difficult to track counterfeits, and leads to insufficient quality control.
In addition, lack of transparency is a serious problem that leads to a lack of traceability of batches. In the nutrition business, this hinders regulatory control, creates risks for consumer safety, and makes effective quality control in nutrition manufacturing impossible. The inability to quickly track and, if necessary, recall a batch leads to significant financial losses.
Difficulties With Audits
If a nutritional business does not implement digital solutions, such as ERP systems, then a standard audit can be difficult. The main challenges include a lack of consistency, insufficient resources and time, and difficulties with data collection and analysis. In a nutrition business, auditing is a complex process that covers product quality, safety, legal requirements compliance, production conditions, and other factors. If a company lacks an automated system, then auditing is perceived by staff as an additional burden, and manual data collection is inefficient and often inaccurate.
Related Service: Odoo DevelopmentHow an ERP System Helps Nutrition Companies Automate Compliance
As we mentioned above, no modern business, including nutrition brands, can develop without a digital ecosystem, namely, ERP solutions. This software helps companies to effectively automate and integrate key business processes, such as accounting, production, supply chain, procurement, and others. In addition, ERP systems help businesses automate regulatory compliance. This is achieved through the automation and standardization of routine processes, transparent and centralized data recording, and the provision of tools for real-time quality monitoring.
Ensuring Full Traceability of Batches
The ERP system automates processes, centralizes data, and provides unique identifiers for each batch. This allows the company to track the movement of goods from the receipt of raw materials to the shipment of finished products. This significantly improves the supply chain and helps control inventory to prevent shortages or surpluses.
Quality Control at Every Production Stage
Production process management is one of the key functions of an ERP system. It monitors product quality at every stage of production, ensuring compliance with standards and customer requirements. This is possible primarily thanks to a single database and, again, process automation. An ERP system helps minimize errors, standardize processes, and significantly increase efficiency.
Simplicity of Auditing and Storage of All Verification Data
An ERP system is a single source of information that also provides analytics and recording functions. In this way, this system transforms a huge amount of unstructured information into graphs and charts. An ERP system stores all financial, managerial, and production history of a business in a verifiable and transparent form. ERP systems enable auditing to function as a routine check rather than as a chaotic process.
Automatic Tracking of Expiration Dates and Non-Conformities
In the ERP system, all information about goods, including characteristics, movement history, and expiration dates, is updated in real time. The system records every operation, and balances are updated automatically, allowing you to control the movement of goods at all stages. For example, if information about a product’s expiration date is entered into the system when it arrives, this parameter will then be tracked automatically.
Odoo for Nutrition Compliance: What You Need to Know
Odoo is a set of open-source business modules that covers all your company’s needs. Odoo solutions are used to comply with food regulations, such as recipe management, product labeling, legal compliance, and food safety control. Odoo enables nutrition brands to automate processes and ensure compliance with laws and standards.
Odoo Quality Module
This module is a quality management tool that helps identify and eliminate the causes of defects, thereby contributing to an overall improvement in the quality of products and services. This module allows you to guarantee product quality not only at the production stage but also during inventory movement. The system supports both scheduled and random checks, thus ensuring quality control at all stages of the process.
The Quality Module provides flexible settings for the frequency of checks, allowing processes to be adapted to the company’s specific needs. In addition, Odoo supports several types of quality checks:
- Checking instructions
- Product snapshot for visual inspection
- Checking product compliance with established criteria
- Recording physical product parameters and assessing their compliance with standards
If problems or defects are detected, the system automatically generates a quality alert, allowing timely measures to be taken to reduce risks and losses. The Odoo system also simplifies the management of defective products and ensures that they are separated from quality goods. Additionally, the system allows you to set up special storage areas for products that have not passed quality control.
Automatic Document Generation
The Odoo Document Management Module allows you to manage files online from one place and eliminates the need for scanning or manual accounting of invoices. The module lets you automatically create documents in the system, which can be done by processing incoming emails or scanned PDF files. In addition, Odoo documentation management allows you to automatically extract data from such documents and create invoices from suppliers or others. This automation significantly reduces manual data entry.
Integration With Production, Warehouses, and Delivery
Odoo is an excellent solution for optimizing production processes, as this system provides all the capabilities on a single platform. You can manage production processes through the MRP module, which allows you to manage product specifications, create production orders, and track production operations. In addition, Odoo provides a powerful module for warehouse and inventory management. You can create and configure warehouses, track the movement of goods, and conduct inventory. Odoo can also integrate with external logistics companies and allows you to configure different delivery methods and link them to specific carriers.
Thus, Odoo ERP for supplement companies is an indispensable tool that provides control, flexibility, and regulatory compliance.
Related Service: Odoo Integration
Business Results of Odoo ERP Implementation
Minimizing the Risk of Fines
ERP systems, including Odoo ERP, provide automated accounting and reporting, enabling companies to comply with legal and industry standards. Nutrition businesses gain a significant advantage from using Odoo tools for GMP compliance. The system also generates accurate reports promptly, which are important for internal control and necessary for submission to regulatory authorities. Compliance with all standards, requirements, and deadlines allows nutrition companies to avoid fines and make processes as transparent as possible.
Related Case Study: Odoo for GENIUS NUTRITION S.R.LIncreasing Customer Confidence
The ERP system provides prompt problem-solving and personalized service. The system consolidates all customer information, allowing you to respond quickly to inquiries and provide accurate and timely information about orders. Built-in analytics enable you to predict customer preferences and tailor products to demand. With these capabilities, you can provide accurate information and thus strengthen customer trust to create and enhance a positive brand reputation.
Easier Scaling to New Markets
Odoo ERP is extremely flexible thanks to its open-source code, so the system can grow with the company as you scale your business. The ERP system lets you standardize key processes and simplifies their replication in new markets. The system can be configured to comply with other legislation, which is particularly important when entering new markets. ERP also allows you to quickly add new employees and manage their access rights to the system, further simplifying work in a new market.
See How Odoo ERP Can Help Reduce Compliance Risk for Your Nutrition Business
Final Thoughts
Due to Odoo’s flexibility and mobility, it is an excellent software solution for compliance management in the nutrition industry. The system provides a variety of tools for tracking production processes, quality control, and regulatory compliance.
Contact our specialists right now and find out how VentorTech implements Odoo for quality and compliance management in the nutrition business!
Recommended articles:
- Why Odoo Works for Thousands but Failed for You? The Hidden Truth Behind Odoo Complaints
- Will your Odoo Enterprise contract be surcharged 25% in April? Tricky cases explained…
- How to Calculate Odoo Workers for Demanding Systems
- Odoo Implementation Steps
- Odoo Access Rights Structure
- AI Order Processing in Odoo
- Odoo EDI Integration
- Key Performance Indicators (KPIs) and their Role in Successful ERP Implementation
- 10 most common questions to the official Odoo partner
